Two recent publications in the Journal of Applied Toxicology (Géniès et al., 2025a,b) highlight the suitability of Phenion® Full-Thickness (FT) Skin Models for investigating dermal absorption and metabolism of topically applied compounds.
Mar 31, 2026
New Study Confirms Structural and Metabolic Competence of Phenion® FT Skin Models in Skin Permeation and Metabolism Research
In a Cosmetics Europe LLRS-funded study, researchers from Pierre Fabre compared the penetration and metabolic profiles of the phytoestrogens genistein and daidzein—commonly used in anti-aging formulations—across four systems: pig skin, fresh and frozen human skin, and the Phenion® FT Skin Model. These molecules are valued for their antioxidant and skin-protective properties, yet comprehensive data on dermal exposure remain limited.
To address this gap, radio-labelled compounds were applied in different concentrations and vehicles (ethanol and cosmetic formulation). Results showed comparable absorption and penetration across all test systems, including Phenion® FT Skin Models—without the need for correction factors. The authors concluded that Phenion® FT models are a reliable alternative to human skin for assessing dermal absorption under these conditions. Differences in bioavailability and tissue distribution between ethanol and formulation applications were also consistent across all skin types.
Importantly, first-pass metabolites (glucuronide and sulfate conjugates) were detected in all specimens, confirming metabolic competence.
These findings underscore the predictive power of Phenion® FT models for dermal absorption and metabolism studies. With their vivo-like properties they are valuable tools for New Approach Methodologies (NAMs) within the New Generation Risk Assessment (NGRA) framework—supporting the 3R principles of reducing, refining, and replacing animal testing.
Phenion®—where science meets the future of cosmetic safety
Géniès, C., Jeanjean, C., Najjar, A., Schepky, A., Lange, D., Kühnl, J., Fabian, E., Zifle, A., Duplan,
H., Hewitt, N. J., & Jacques, C. (2025a). Characterization of the in vitro penetration and first-pass metabolism of genistein and daidzein using human and pig skin explants and Phenion full-thickness skin models. Journal of Applied Toxicology, 45(2),200–209. https://doi.org/10.1002/jat.4689
Géniès, C., Jeanjean, C., Najjar, A., Schepky, A., Lange, D., Kühnl, J., Fabian, E., Zifle, A., Duplan,
H., Hewitt, N. J., & Jacques, C. (2025b). Effect of vehicle on the in vitro penetration and metabolism of genistein and daidzein in ex vivo skin explants and the Phenion full-thickness skin model. Journal of Applied Toxicology, 45(2), 210–221. https://doi.org/10.1002/jat.4693
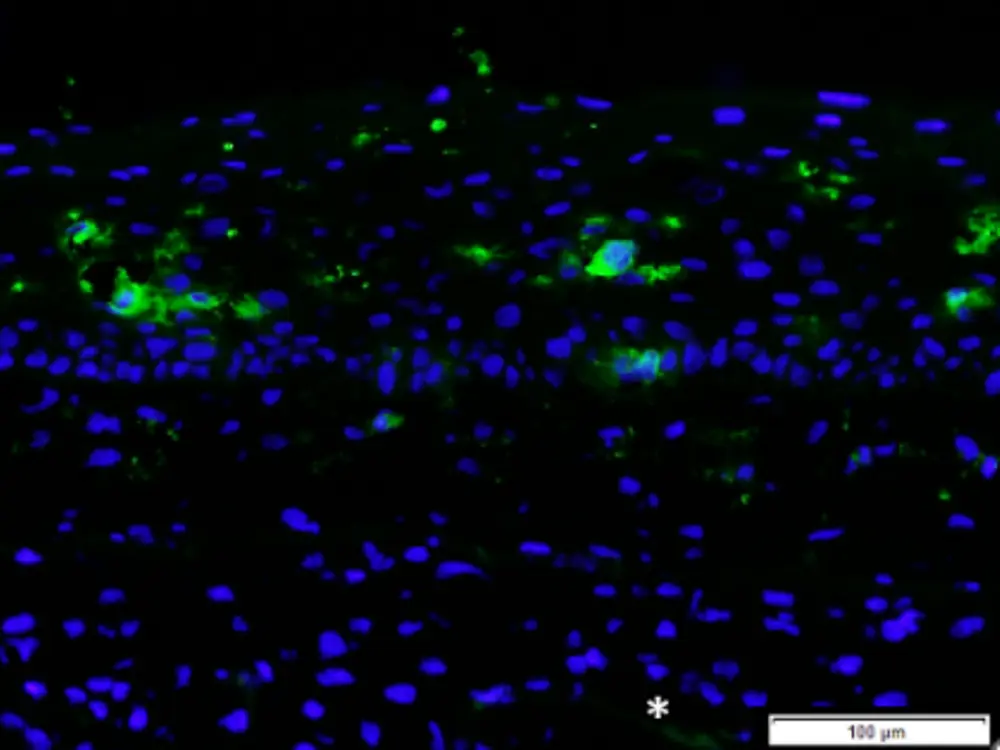
Phenion FT Skin Model with integrated MUTZ-LC’s, labelled for CD1a (green fluorescent cells in the epidermis; cells counterstained