The latest study by Böttcher et al. (TIV 2025) demonstrates a major step forward in developing immunocompetent in vitro models for skin sensitization testing—an essential component of next generation risk assessment (NGRA) aligned with the EU Cosmetics Regulation and OECD test guidelines. The research successfully integrates MUTZ-derived Langerhans cells (MUTZ-LCs) into the Phenion® Full-Thickness (FT) Skin Model, creating a physiologically relevant system that reflects key events of the skin sensitization Adverse Outcome Pathway (AOP).
Apr 6, 2026
Breakthrough in Immunocompetent Skin Models: MUTZ-LCs Successfully Integrated into Phenion® Full-Thickness Skin Equivalent
Unlike conventional New Approach Methodologies (NAMs) for skin sensitization testing, which often lack immune cell components, this innovative approach combines keratinocytes, fibroblasts, and functional dendritic cells in a single 3D model.
Langerhans cells are key players in allergic contact dermatitis, orchestrating immune responses upon exposure to sensitizers. Until now, their inclusion in reconstructed skin models has been limited by challenges in cell differentiation and culture standardization. Our latest research demonstrates that MUTZ-3 cells can be efficiently differentiated into LC-like cells under optimized conditions—reducing fetal calf serum (FCS) content from 20% to just 5% and eliminating the need for undefined supplements such as 5637-conditioned medium. This refinement not only accelerates differentiation from 14 to 7 days without functional loss but also aligns with the 3R principles by minimizing animal-derived components.
The optimized MUTZ-LCs exhibit key markers of native LCs, including CD1a and CD207 (langerin), and respond to sensitizers like DNCB with increased CD83 expression, confirming their functional competence. Most importantly, these cells were successfully integrated into the Phenion® FT Skin Model, distributing evenly within the epidermis and maintaining their dendritic morphology—creating a physiologically relevant platform for assessing skin sensitization potential.
This advancement paves the way for more standardized, cost-efficient, and ethically responsible testing strategies. By combining keratinocytes, fibroblasts, and immune cells in a single system, Phenion brings the industry closer to a stand-alone in vitro method that addresses multiple key events of the OECD AOP for skin sensitization.
Phenion®—Innovating for a future without animal testing.
Phenion FT Skin Model with integrated MUTZ-LC’s, labelled for CD1a (green fluorescent cells in the epidermis; cells counterstained with DAPI, blue fluorescence)
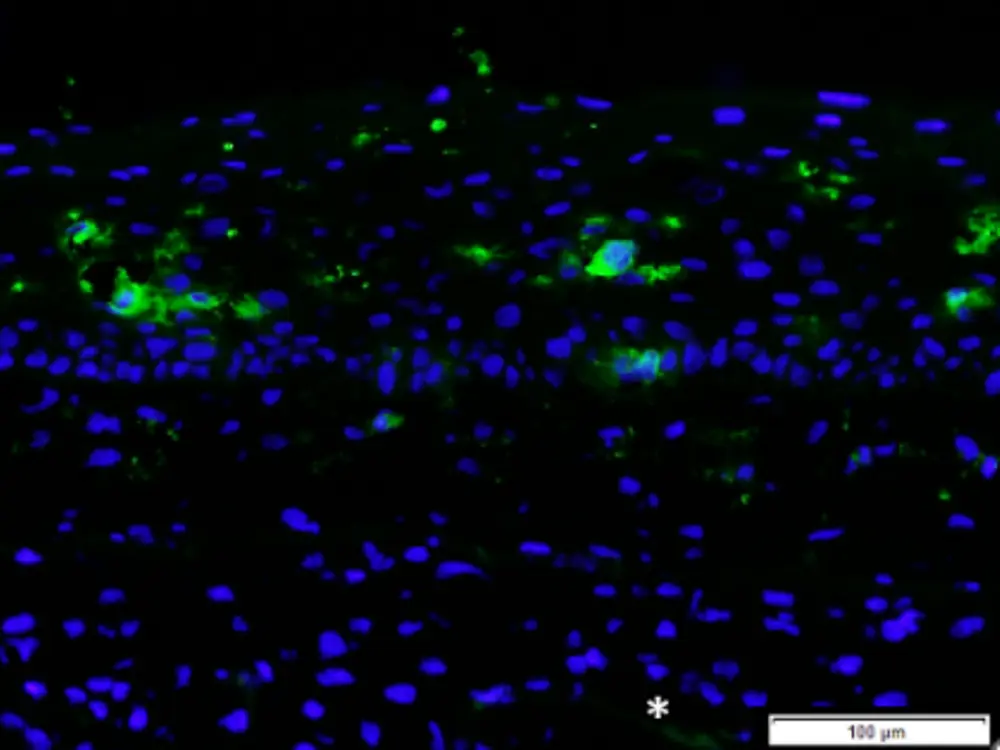
Phenion FT Skin Model with integrated MUTZ-LC’s, labelled for CD1a (green fluorescent cells in the epidermis; cells counterstained